I. How to Use
When to Use
CARE-based risk stratification may be applied to all adult patients over eighteen years of age undergoing cardiac surgery. However, it is important to note that the score was not validated for ventricular assist device insertion or heart transplantation, as these procedures were excluded from the original study design.
Pearls / Pitfalls
The CARE score is grounded in the well-established understanding that clinicians are generally reliable at identifying patients at the extremes of cardiac surgical risk, those who are clearly low risk (CARE 1) and those who are unequivocally very high risk (CARE 5). In contrast, determining intermediate levels of risk is more complex and more susceptible to variability when based solely on subjective clinical judgment.
To reduce this inconsistency, the CARE system incorporates two objective categories of risk factors- surgical complexity and the presence of comorbid conditions- which help define the intermediate tiers of CARE 2 through CARE 4. This structure reflects the rationale that patients with controlled medical conditions (CARE 2) face greater risk than those without any comorbid disease (CARE 1), yet remain at lower risk than patients with uncontrolled conditions (CARE 3). CARE 3 further acknowledges that uncontrolled comorbidities confer a level of risk comparable to that associated with complex surgical procedures, a relationship supported by multiple regression analyses. Because of this similarity, both factors are assigned equal prognostic weight. CARE 4 represents the additive burden that arises when complex surgery coincides with uncontrolled medical disease.
Emergency surgery receives special designation within the system, as catastrophic physiologic states are among the most powerful predictors of adverse cardiac surgery outcomes. Importantly, CARE categories 1 and 2 are ineligible for this emergency classification. The score’s use of a small number of robust, clearly defined variables enhances its stability and reliability as a risk-adjusted predictor of morbidity.
Major morbidity within the CARE framework encompasses several postoperative complications. Cardiovascular morbidity includes low cardiac output and hypotension requiring an intra-aortic balloon pump or prolonged use of multiple vasopressors or inotropes, as well as malignant arrhythmias requiring cardiopulmonary resuscitation, antiarrhythmic therapy, or device intervention. Respiratory complications include mechanical ventilation beyond 48 hours, tracheostomy, or the need for reintubation. Neurologic morbidity encompasses focal brain injury with permanent deficits or irreversible encephalopathy. Renal morbidity is defined by acute renal failure requiring dialysis. Infectious complications include septic shock with positive blood cultures or deep sternal or leg wound infections necessitating antibiotics and/or surgical debridement. Finally, a miscellaneous category captures any secondary surgery or invasive procedure required to treat complications arising from the initial cardiac operation.
Why Use
Complex perioperative risk tools such as the Society of Thoracic Surgeons (STS) score, the European System for Cardiac Operative Risk Evaluation (EuroSCORE), EuroSCORE II, and the Parsonnet score incorporate numerous clinical variables, which can make them cumbersome and may limit routine use in everyday practice. Conversely, clinical judgment supplemented by a small number of well-established risk factors has been shown to provide practical and reasonably accurate prognostic information. The ordinal CARE score was created with this streamlined approach in mind. It can be applied quickly across a wide range of clinical situations without requiring extensive data collection, and its structure is reminiscent of the American Society of Anesthesiologists (ASA) physical status classification, a system already familiar to perioperative clinicians.
II. Next Steps
Advice
The CARE score can complement other risk indices and clinical judgment to support perioperative planning, resource allocation, and patient counseling. Patients flagged as intermediate to high-risk by the CARE score may warrant closer monitoring, targeted optimization of comorbid conditions, and coordinated multidisciplinary planning. However, the score should be implemented as an adjunct and not as a substitute for a thorough clinical evaluation.
III. Evidence
Evidence Appraisal
CARE provides discrimination and calibration for predicting mortality and major morbidity after cardiac surgery that is comparable to widely used models such as EuroSCORE and the STS score. However, it differs in important ways, particularly in its relative simplicity, generalizability, and breadth of its validation. Although mortality after cardiac surgery has declined substantially over the past two decades largely because of advances in surgical technique, the CARE score has continued to demonstrate stable calibration without requiring frequent readjustment. One recognized limitation, however, is that it tends to overestimate perioperative risk in patients who fall into the very high-risk category.
Unlike EuroSCORE and STS, which have undergone extensive multicenter validation, the CARE score was developed primarily from a single-center national cohort. This more limited scope likely contributes to its lower prevalence in clinical practice. STS, on the other hand, enjoys widespread use, notably in the United States, and both STS and EuroSCORE have the added advantage of being adaptable for benchmarking, research, and multicenter comparisons- capabilities not yet established for CARE.
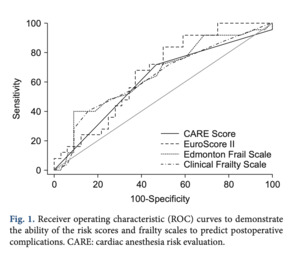
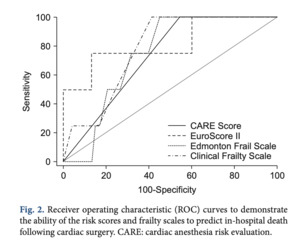
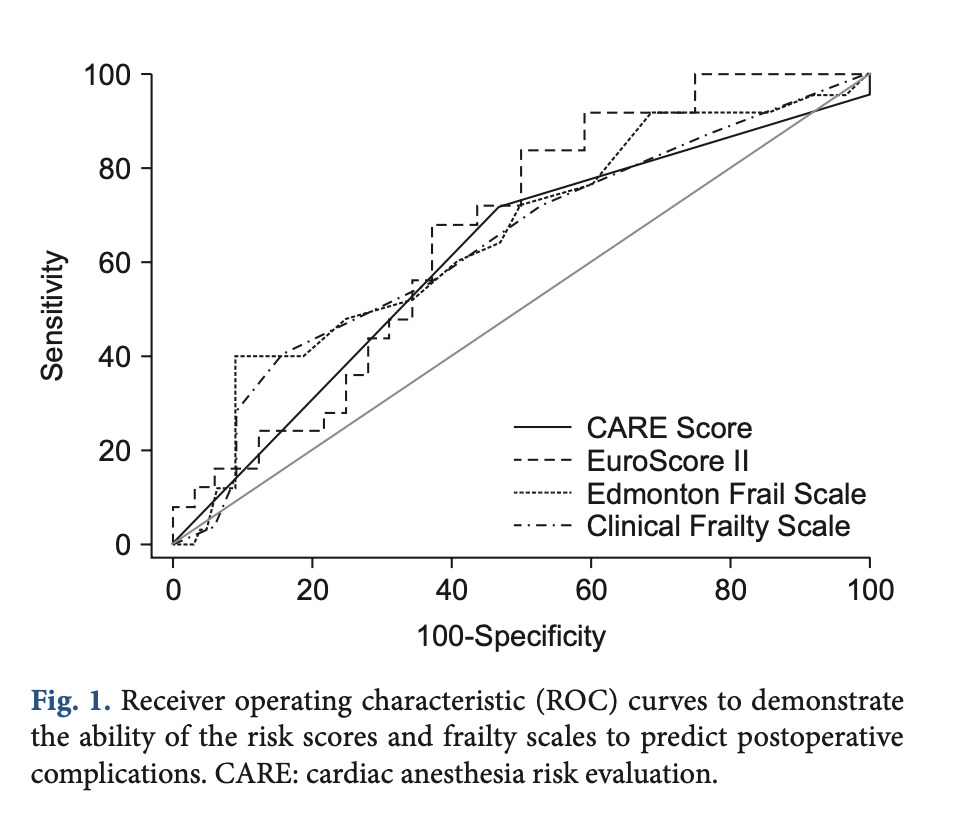
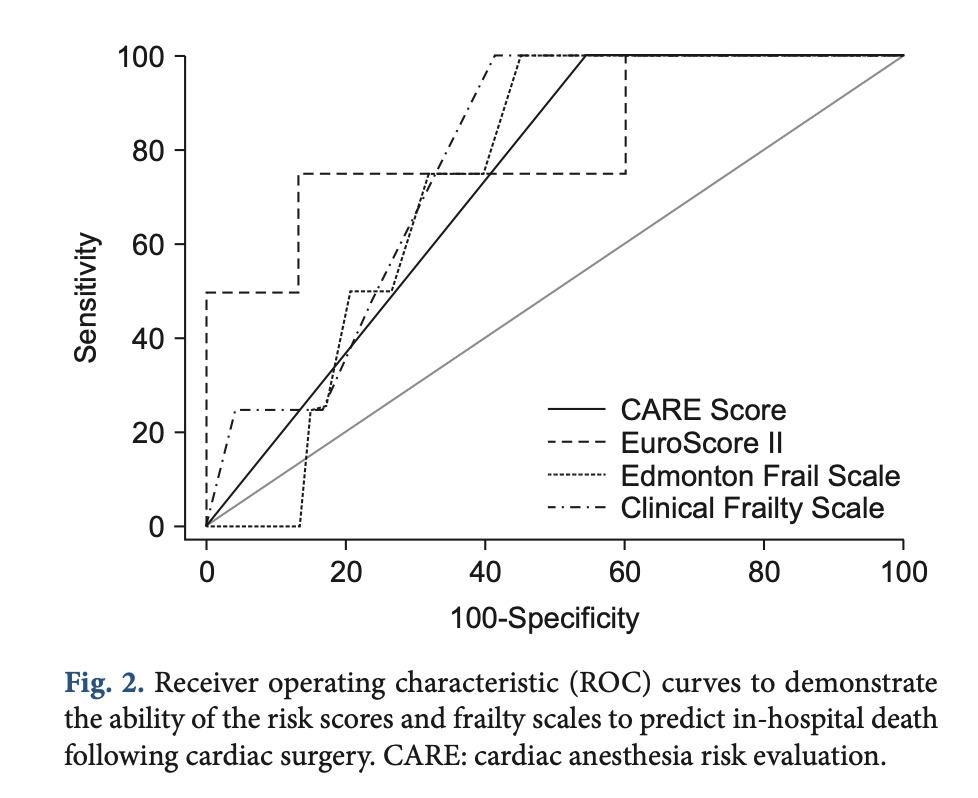
Despite these constraints, several additional applications of the CARE score have been explored in the literature. Bhukal and colleagues found that while CARE was a good predictor of mortality, it performed poorly in forecasting hospital length of stay. Similarly, Wagener et al. reported that although CARE predicted mortality almost as effectively as the Surgical Procedure Assessment (SPA) score, it did not reliably predict ICU length of stay. Comparisons with frailty measures such as the Edmonton Frailty Scale and the Clinical Frailty Scale revealed that CARE was less effective in predicting in-hospital mortality, length of stay, and postoperative complications.
Further insights emerged from Ristovic et al. who examined intraoperative hypotension among intermediate-risk patients categorized as CARE 3 or 4. In this group, mortality increased by 18% for every additional 10 minutes during which mean arterial pressure (MAP) remained between 55 and 64 mmHg after cardiopulmonary bypass, and by 30% for every 10 minutes with MAP below 55 mmHg. Interestingly, this association did not hold for CARE 5 patients, in whom intraoperative hypotension did not correlate with higher mortality.
Together, these findings highlight both the strengths and limitations of the CARE score, underscoring its utility as a streamlined risk assessment tool while also emphasizing areas where broader validation and further study are needed.
Formula
(Unchanged since 2001)
CARE 1: patient with stable cardiac disease and no other medical problems, undergoing non-complex surgery
CARE 2: patient with stable cardiac disease and one or more controlled medical problems, undergoing noncomplex surgery
CARE 3: patient with any uncontrolled medical problem OR a patient undergoing complex surgery
CARE 4: patient with any uncontrolled medical problem AND undergoing complex surgery
CARE 5: patient with chronic or advanced cardiac disease for whom cardiac surgery is undertaken as a last hope to save or improve life
E (only applicable to CARE 3-5): surgery as soon as diagnosis is made and operating room is available
Facts & Figures
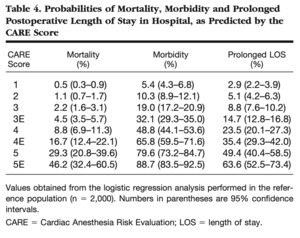
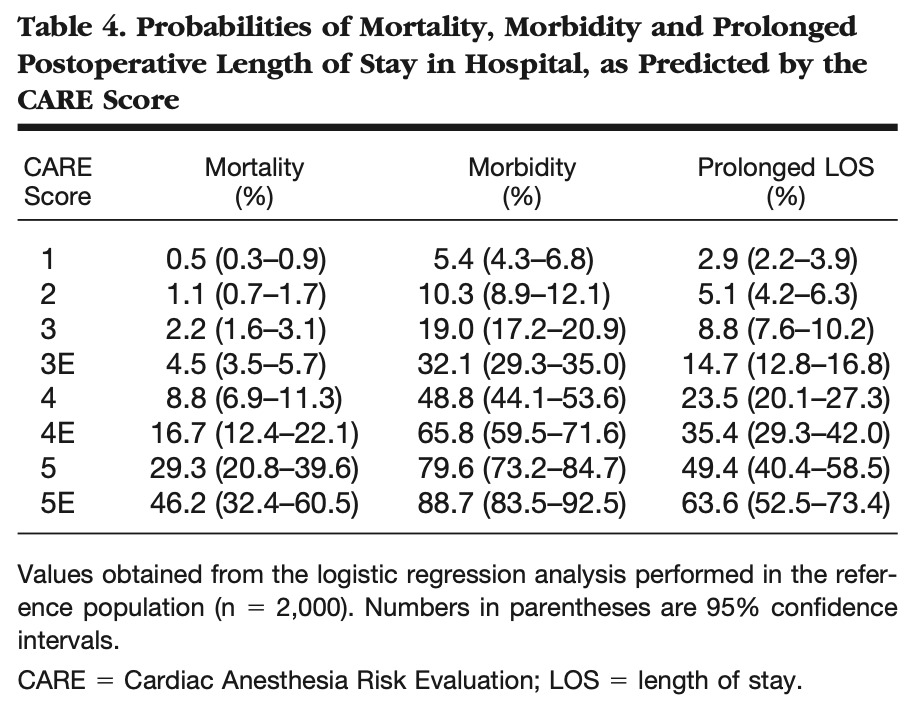
Description of mortality and morbidity rates based on CARE (Dupuis 2001)
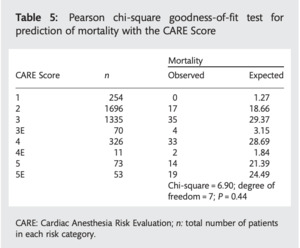
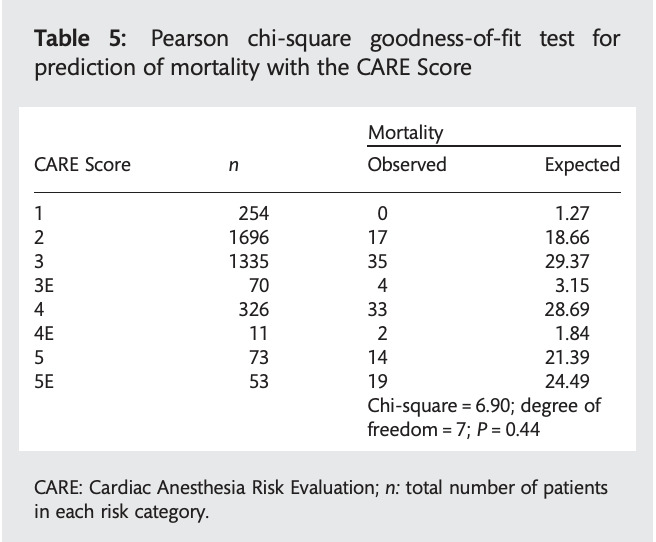
Demonstration of CARE calibration (Tran 2012)
Areas under the Receiver Operating Characteristic (ROC) curves for the CARE score were approximately 0.615 and 0.726 for postoperative complications and in-hospital mortality, respectively, demonstrating relatively low probability to predict outcomes (Kovacs 2017)
Literature
Dupuis JY, Wang F, Nathan H, Lam M, Grimes S, Bourke M. The Cardiac Anesthesia Risk Evaluation Score: A Clinically Useful Predictor of Mortality and Morbidity after Cardiac Surgery. Anesthesiology. 2001;94(2):194
Tran DTT, Dupuis JY, Mesana T, Ruel M, Nathan HJ. Comparison of the EuroSCORE and Cardiac Anesthesia Risk Evaluation (CARE) score for risk-adjusted mortality analysis in cardiac surgery. Eur J Cardio-Thorac Surg Off J Eur Assoc Cardio-Thorac Surg. 2012;41(2):307-313
Pittams AP, Iddawela S, Zaidi S, Tyson N, Harky A. Scoring Systems for Risk Stratification in Patients Undergoing Cardiac Surgery. Journal of Cardiothoracic and Vascular Anesthesia. 2022;36(4)
Bhukal I, Solanki SL, Ramaswamy S, Yaddanapudi LN, Jain A, Kumar P. Perioperative Predictors of Morbidity and Mortality Following Cardiac Surgery under Cardiopulmonary Bypass. Saudi J Anaesth. 2012;6(3):242-247
Wagener G, Minhaz M, Wang S, et al. The Surgical Procedure Assessment (SPA) Score Predicts Intensive Care Unit Length of Stay after Cardiac Surgery. J Thorac Cardiovasc Surg. 2011;142(2);443-350
Ad N, Homes SD, Patel J, Pritchard G, Shuman DJ, Halpin L. Comparison of EuroSCORE II, Original EuroSCORE, and the Society of Thoracic Surgeons Risk Score in Cardiac Surgery Patients. Ann Thorac Surg. 2016;102(2):573-579.
Ouattara A, Niculescu M, Ghazouani S, et al. Predictive Performance and Variability of the Cardiac Anesthesia Risk Evaluation Score. Anesthesiology. 2004;100(6):1405-1410.
Kovacs J, Moraru L, Antal K, Cioc A, Voidazan S, Szabo A. Are Frailty Scales Better than Anesthesia or Surgical Scales to Determine Risk in Cardiac Surgery? Korean J Anesthesiol. 2017;70(2):157-162.
Ristovic V, de Roock S, Mesana TG, van Diepen S, Sun LY. The Impact of Preoperative Risk on the Association between Hypotension and Mortality after Cardiac Surgery: An Observational Study. J Clin Med. 2020;9(7): 2057